Welcome to HortHerb Publisher
 Harnessing Genetic Populations in Plant Breedi...
Harnessing Genetic Populations in Plant Breedi... The Fruit Revolution in the Genomic Era: Unvei...
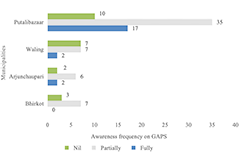
The Fruit Revolution in the Genomic Era: Unvei... The Role of Genome-Wide Association Studies (G...
The Role of Genome-Wide Association Studies (G... ECONOMICS OF PRODUCTION AND MARKETING FOR FREN...
ECONOMICS OF PRODUCTION AND MARKETING FOR FREN... Optimizing Chickpea ( Cicer arietinum L.) Yie...
Optimizing Chickpea ( Cicer arietinum L.) Yie... Economic Analysis of Apple Production and Mark...
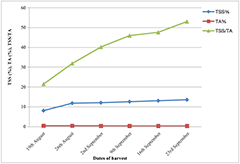
Economic Analysis of Apple Production and Mark... Effect of Different Postharvest Treatments on ...
Effect of Different Postharvest Treatments on ... Productivity Determinants and Production Const...
Productivity Determinants and Production Const...
- A research team from the Chinese Academy of Science...(14 Mar.2024)
- A research team from the Institute of Botany of the ...(14 Mar.2024)
- A research team led by Professor Li Chenlong from S...(01 Mar.2024)
- A research team led by Dr. Zhang Yangyong from the I...(01 Mar.2024)